 The atomic radius in the periodic table decreases across the period and increases down the group.Ītomic radius decreases across the period The bigger the atomic size, the larger the atomic radius. Then depending on atomic size, the atomic radius of the two atoms are calculated. So to calculate the atomic radius, at first the distance between the nuclei is measured. In the same way the anions can have lesser radius that the radius of its original neutral atom.The ionic radius is thus reduced than atomic radius. However the nucleus attract the rest of the electrons more tightly towards the nucleus. When an atom loses electron sometime it has less orbital than before.The atom can be cation or anion by losing or gaining electron. Again the atomic radius is different because of their ionic status of the atom.The atoms of different element, they must differ in electrons and protons, sometimes number of orbitals and so on.Here the radius of two ions are not equal and few other things need to be discussed before we calculate the ionic radius: Ionic radius is the radius calculated when an atom is bonded with another atom in a molecule by transferring electrons to make ionic bond. The metallic diameter is equal to the distance between two nuclei and the radius is just half of it. The metallic radius can also be calculated by measuring the distance between the two nuclei of the two atoms. The metallic radius is the radius calculated between two metal atoms bonded together in metal cluster. The diameter of an atom is equal to the distance between two nuclei and the atomic radius will be the half of it. When an atom (A) is covalently bonded with another homonuclear atom (A), the atomic radius of that atom can be calculated by measuring the distance between the two nucleus of those two atoms. Covalent radiusĬovalent radius is the radius calculated when an atom is bonded covalently with another atom of same element. 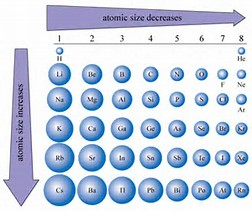
There are different methods of calculating the radius by measuring the distance between two nucleus when an atom is bonded in a molecule. So, as the position of electron is not certain rather they are explained as electron cloud around the nucleus, it is hard to measure an atomic radius accurately. Ways to measure the atomic radiusĪccording to the Heisenberg uncertainty principle, it is not possible to measure the momentum and the position of the electron simultaneously. 
Thus sometime different methods are used to measure the radius while they are bonded in a molecule. As there are no physical existence of orbital in atoms, it is difficult to measure the atomic radius. Write Electronic Structure of Bromine.The atomic radius is the size of the atom, typically measured by the distance from the nucleus of the atom to the electron clouds around the nucleus. 
Show Electronic configuration of Bromine. On which block Bromine element is present? It is also use to make fireproof circuit board. It is also use in agriculture to make fumigant It is mostly use in medicine for the treatment of drug addicted personīromine is also use in water purification process. Reactivity : Bromine is reactive than iodine (I) and less than fluorine (F) and chlorine (Cl)īromine is use in pharmaceutical industryīromine is also use in electronic device. 
In outermost shell 1 electron is required to complete their octateĪtomic radius : atomic radius of Bromine is 94 Pm Įlectronic configuration of Bromine is 4S 23d 10 4P 5Įlectronic configuration in concert of shell 2,8,18,7 In this isotope 35 proton, 35 electron and 49 neutron is present. In this isotope 35 proton,35 electron and 44 neutron is presentĨ1Br: atomic weight of this isotope is 80.9 Amu Melting point :melting point of bromine is – 7.2 0 Cħ9Br: atomic weight of this isotope is 78.9 Position : bromine is place in periodic table at 4 thperiod ( row) and 17 th column ( group)īoiling point :boiling point of bromine is 58.8 0C In bromine 35 proton and 45 electron is present In bromine 35 proton and 35 electron is presentĪtomic weight/ mass: atomic weight of bromine is 79.9 approximately 80 Amu The ne is originated from Greek word ‘ Bromis’ meaning of this word is stenchĪtomic symbol: atomic symbol of bromine is BrĪtomic number : atomic number of bromine is 35 ĭiscovery : it is discovered in 1826 by scientist Antoine serome Belard and Carl lowing It is mostly found in sea water in shell fish. Due to toxic nature it’s harmful to body like irritation in eyes and effect on throat also irritation for skin. Bromine is toxic in nature and gives sharp smell. Bromine place in halogen family and lie properties in between chlorine and iodine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
